Collagen peptide preparation techniques include chemical methods, enzymatic methods, thermal degradation methods and a combination of these methods. The molecular weight range of collagen peptides prepared by different techniques varies greatly, with chemical and thermal degradation methods mostly used for the preparation of gelatin and enzymatic methods mostly used for the preparation of collagen peptides.
First generation: chemical hydrolysis method
Using animal skin and bone as raw materials, collagen is hydrolyzed into amino acids and small peptides under acid or alkaline conditions, the reaction conditions are violent, the amino acids are seriously damaged during the production process, L-amino acids are easily converted into D-amino acids and toxic substances such as chloropropanol are formed, and it is difficult to control the hydrolysis process according to the prescribed degree of hydrolysis, this technology has been rarely used in the field of collagen peptides.
The second generation: biological enzymatic method
Using animal skin and bone as raw materials, collagen is hydrolyzed into small peptides under the catalyst of biological enzymes, the reaction conditions are mild and no harmful by-products are generated during the production process, but the molecular weight of the hydrolyzed peptides has a wide range of distribution and uneven molecular weight. this method was more commonly used in the field of collagen peptide preparation before 2010.
The third generation: biological enzymatic digestion + membrane separation method
Using animal skin and bone as raw materials, collagen is hydrolyzed into small peptides under the catalyst of protein hydrolase, and then the molecular weight distribution is controlled by membrane filtration; the reaction conditions are mild, no harmful by-products are generated during the production process, and the product peptides have narrow molecular weight distribution and controllable molecular weight; this technology was applied one after another around 2015.
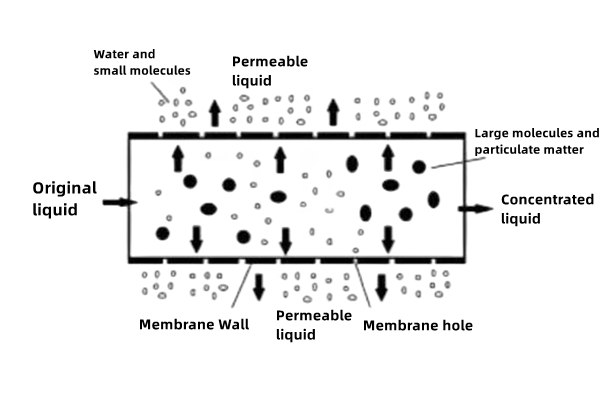
Fourth generation: peptide preparation technology separated by collagen extraction and enzymatic process
Based on the study of thermal stability of collagen, collagen is extracted near the critical thermal denaturation temperature, and the extracted collagen is enzymatically digested by biological enzymes, and then the distribution of molecular weight is controlled by membrane filtration. The temperature control was used to achieve the collagen extraction process invariance, reduce the occurrence of merad reaction and inhibit the formation of colored substances. The reaction conditions are mild, the molecular weight of peptide is uniform and the range is controllable, and it can reduce the generation of volatile substances and inhibit fishy smell, which is the most advanced collagen peptide preparation process until 2019.

Post time: Jan-14-2023

